Potential New Drug Target Could Boost Effectiveness of Chemotherapy Drugs
Researchers at The University of Texas at Austin have discovered that a large family of reverse transcriptases (RTs)—enzymes that are found in all organisms and have been extensively studied for more than 50 years—have the previously unsuspected ability to repair DNA damage. The discovery makes them a potential new drug target that might be exploited to block cancer cells from developing resistance to radiation and chemotherapy drugs. The findings were published today in the journal Cell.
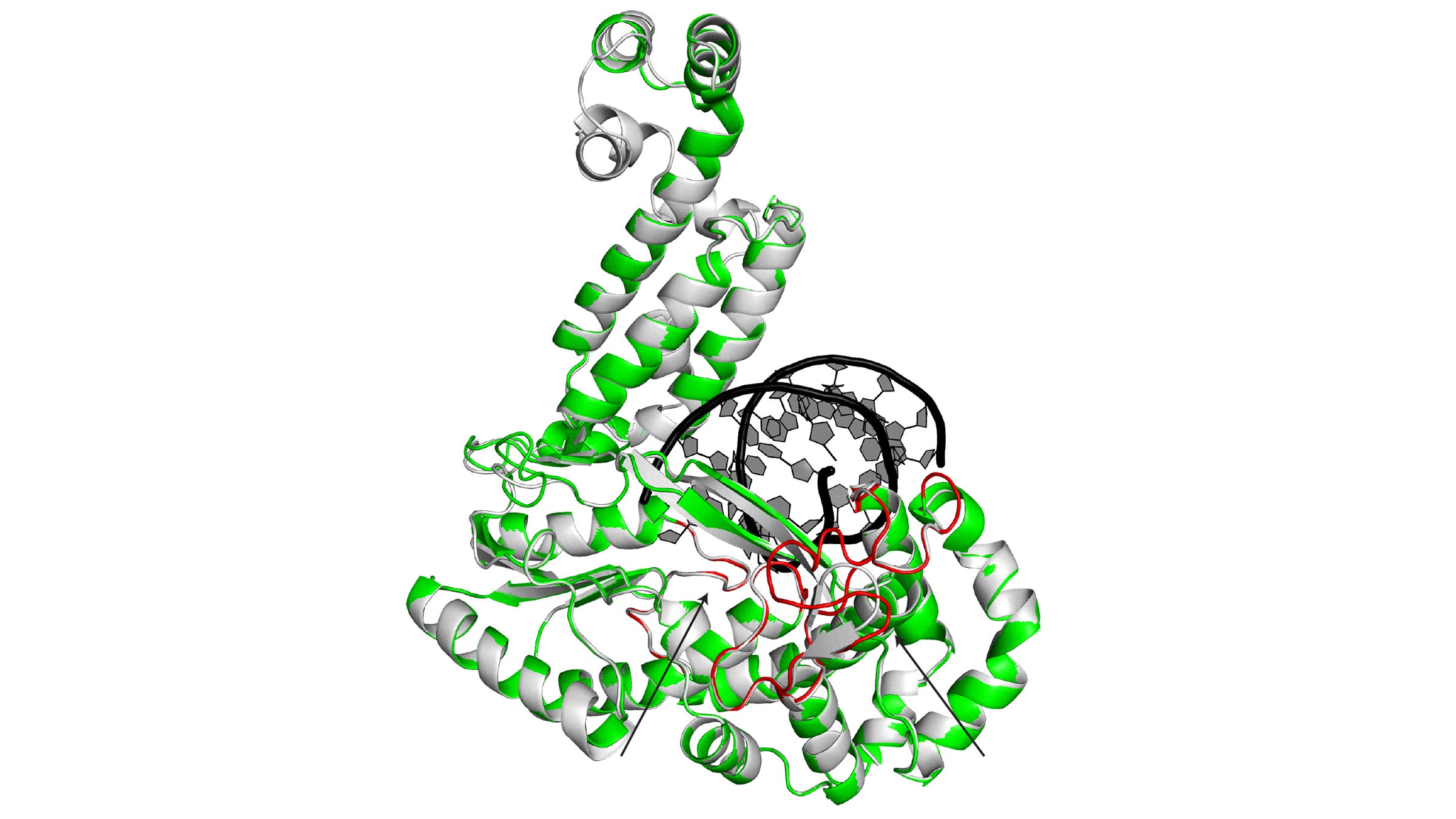
Enzymes in a large family called group II intron-like RTs have 3D structures that are remarkably similar, which suggests they share the ability to help repair double-strand DNA breaks. This image is a superposition of two of these enzymes: G2L4 and GsI-IIC RT. Their shared (or conserved) structures are in alternating green and gray. Credit: University of Texas at Austin.
A research team led by Alan Lambowitz, a professor of molecular biosciences, has discovered that bacteria use enzymes from a large family called group II intron-like RTs to repair double-strand DNA breaks, a lethal form of DNA damage. The three-dimensional structure of these enzymes, including regions that function to repair DNA damage, is virtually identical for all members of this family of reverse transcriptases, including enzymes in human cells encoded by LINE-1 elements.
“These virtually identical structures make it a good bet that these RTs are doing the same thing in humans as they are in bacteria,” Lambowitz said.
In other words, if you know the shape and composition of an object (say, a knife) and you know its function (cutting things), the next time you see an object with the exact same composition and shape, you know it probably does the same thing.
Radiation and some chemotherapy drugs work by damaging the DNA of cancer cells. But given enough time, cancers often develop resistance to these therapeutics by co-opting DNA repair mechanisms that normally help protect healthy human cells from life’s wears and tears.
“We knew about other mechanisms for repairing double-strand breaks in humans,” said Lambowitz. But, he noted, the notion that reverse transcriptase enzymes in human cells could be acting directly to repair double-strand breaks was not known or even suspected. “That’s an entirely novel idea.”
Drug companies are already testing inhibitors that target a previously known human DNA repair enzyme called DNA polymerase theta (Pol theta). But Lambowitz's group has found that there's a whole other class of enzymes that is likely doing the same thing as Pol theta that they didn't even know about. To have an effective chemotherapeutic drug, you would have to knock out all of these systems that are in place to repair DNA breaks induced by chemotherapeutic drugs.
Lambowitz is currently in discussions with other researchers about designing inhibitors based on the predicted structure of human LINE-1 element RTs, as well as genome-editing applications of these enzymes.
Reverse transcriptases were already known to copy RNA sequences into DNA sequences and play critical roles in the replication of both retroviruses, such as HIV-1, as well as mobile genetic elements, which are a type of genetic material that can move around within a genome and are present in bacteria and in hundreds of thousands of copies in the human genome. This latest discovery for the first time adds DNA repair to the list of functions of reverse transcriptases.
Lambowitz and his team have long studied group II introns and closely related enzymes of the same family, referred to as group II intron-like or non-LTR-retroelement reverse transcriptases.
Most of the research in the paper was done by graduate student Seung Kuk Park as part of his Ph.D. thesis. The other authors on the Cell paper are Georg Mohr, Jun Yao and Rick Russell.
Lambowitz has worked with the Office of Technology Commercialization at UT Austin to build a portfolio of intellectual property around the diverse and valuable uses of and optimized designs for group II intron and group II intron-like reverse transcriptases.
Funding for this research was provided by the Welch Foundation and the National Institutes of Health.
The University of Texas at Austin is committed to transparency and disclosure of all potential conflicts of interest and acknowledges the author involved in this study holds intellectual property rights that may yield revenue related to findings described in this article.