Live Cell Imaging Reveals New Clues About Processes Linked to Birth Defects
John Wallingford, professor of molecular biosciences at The University of Texas at Austin, and his team used a process called live cell imaging to make observations about how a developing embryo transforms from its early ball shape into a more elongated shape with a distinct head and rear. Disruptions to this process in human embryos can lead to birth defects.
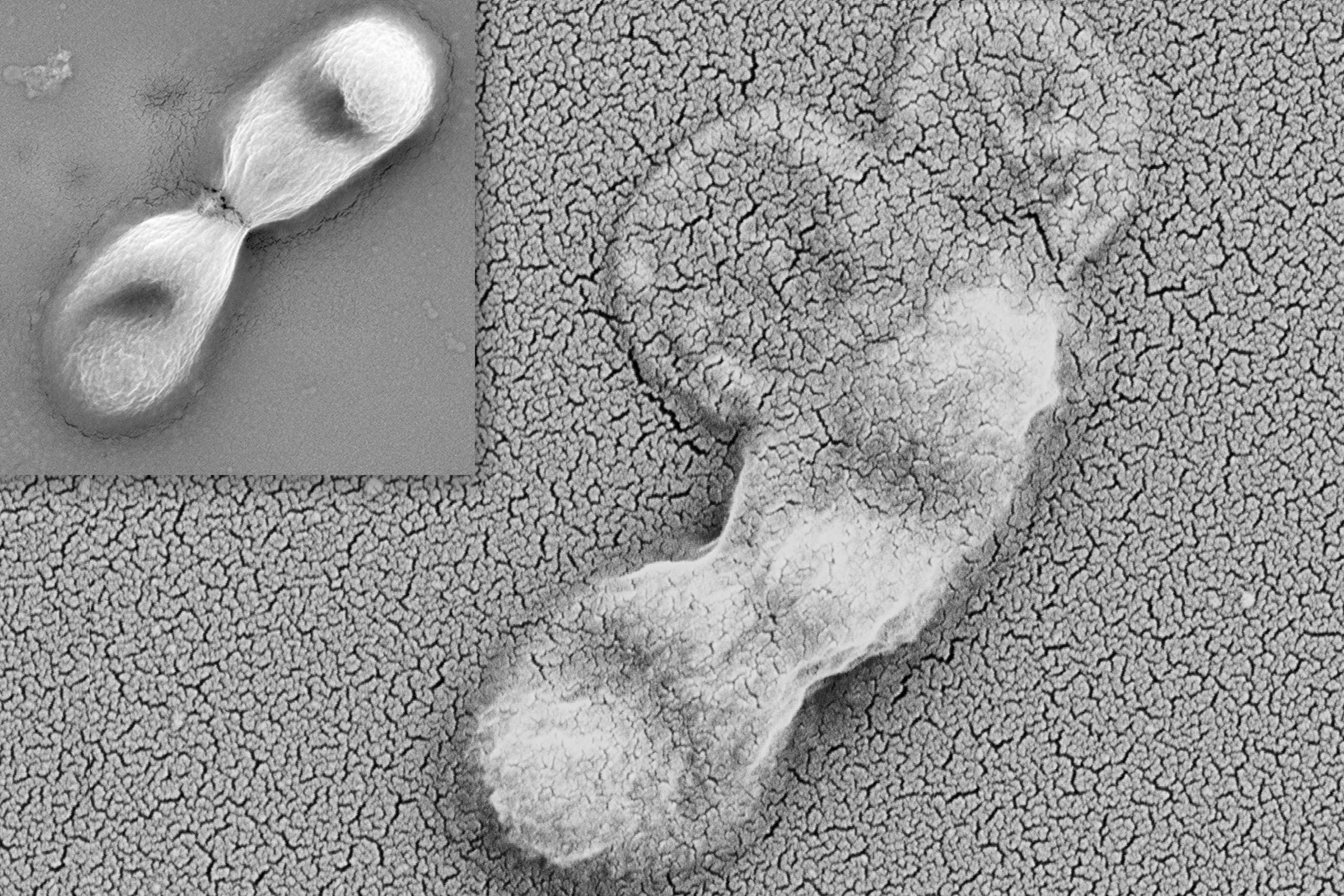
The scientists witnessed in frog embryo cells the flattening and stretching that leads up to an embryo's transformation into a tadpole. They also identified a specific protein that is key to the physical forces driving the process.
In humans, defects in this process cause spina bifida and other more severe neural tube defects (NTDs). NTDs are the second most common human birth defect. Although birth defects receive far less attention than pediatric cancer, in the U.S. they are more than twice as lethal.
"The fascinating thing about embryos is that they construct themselves," Wallingford said. "That is, your own cells built your body when you were an embryo, which is rather like a bunch of bricks building a house. Just as you have to exert some effort to mold a piece of modeling clay, cells in an embryo must generate physical forces to build the embryo."
In this video , the frog cells are starting to flatten from a rounded shape to a horizontally elongated shape. Later, this will allow the cells to stack up like pancakes.
"It's counter-intuitive, but by elongating horizontally, these cells can later move between one another horizontally, which allows them to 'stack up' vertically, which elongates the tissue in the vertical direction," Wallingford said.
In a pair of papers publishing this week in the journals Developmental Cell and Cell Reports, two postdoctoral scholars working with Wallingford, Robert Huebner and Shinuo Weng, explore how a single obscure protein called Arvcf couples the movements of individual cells with larger forces generated by entire tissues in elongating embryos. Without this protein, cells are slower at "intercalating," or stacking up, and tissues can't exert enough force to elongate.
To make the video, the scientists harvested tissue from a frog embryo and dyed the cell membranes with a fluorescent protein. They placed the cells growing in a dish under a microscope and took pictures every few seconds for about an hour.
"Frog embryos normally develop in pond water, as anyone who's watched tadpoles growing knows," Wallingford said. "This makes the frog embryo a great system for studying vertebrate development, as it's fairly straightforward to grow them in a dish of artificial pond water."
A developmental biologist, Wallingford was recently awarded a prestigious Guggenheim Fellowship and is authoring a popular science book about how to research, treat, and prevent birth defects and celebrate human-body variability, according to findings from scientists, medical professionals, patient advocates, disabilities scholars and historians.